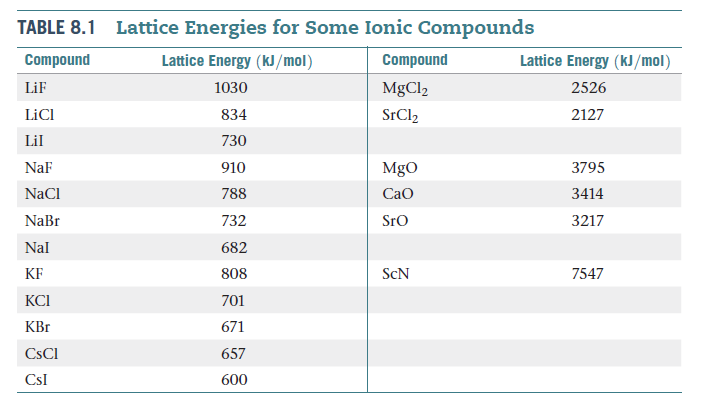
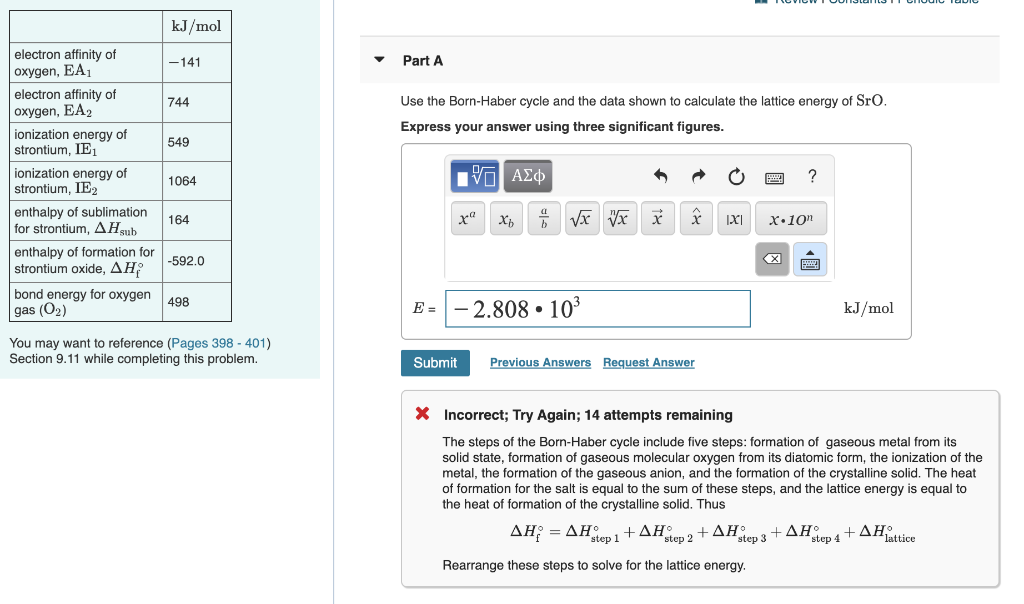
SOLVED: Select the compound with the greatest (i.e, most negative) lattice energy: O A NaF B. Csl C. CaBr2 D. SrO E: BaSO4 Reset Selection Mark for Review What's This?

Amazon.com : Monster Energy Ultra Watermelon, Sugar Free Energy Drink, 16 Ounce (Pack of 24) : Grocery & Gourmet Food

SOLVED: Arrange these substances in Order Of increasing magnitude of lattice energy: KCI, SrO, RbBr, CaO SrO < CaO < KCI < RbBr SrO CaO < RbBr KCI KCI < RbBr <

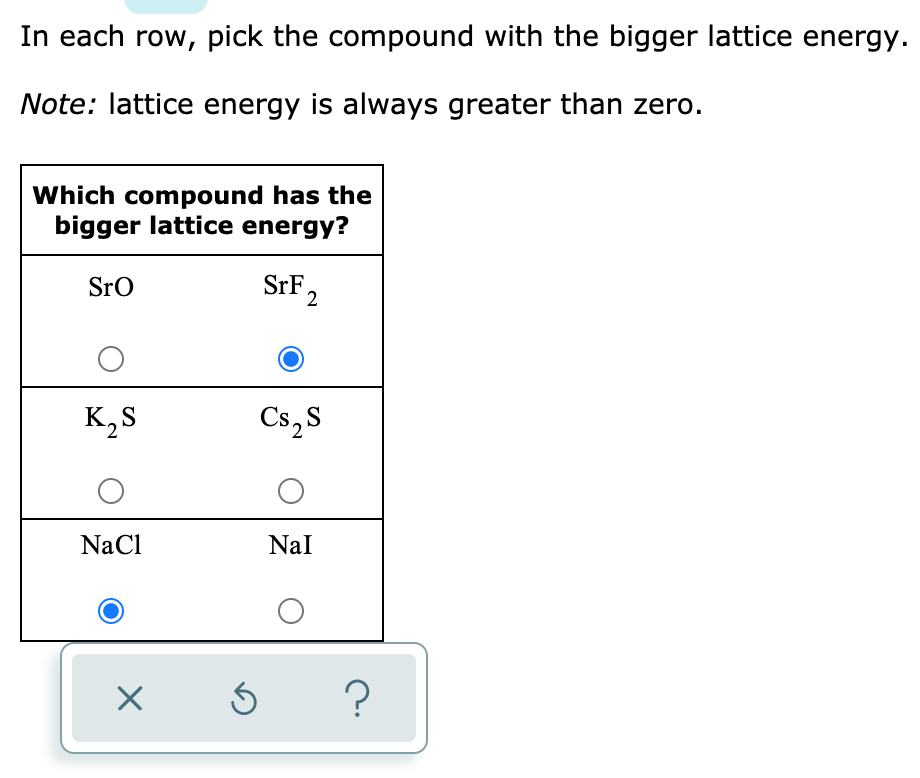
OneClass: In each row, pick the compound with the bigger lattice energy. Note: lattice energy is alwa...
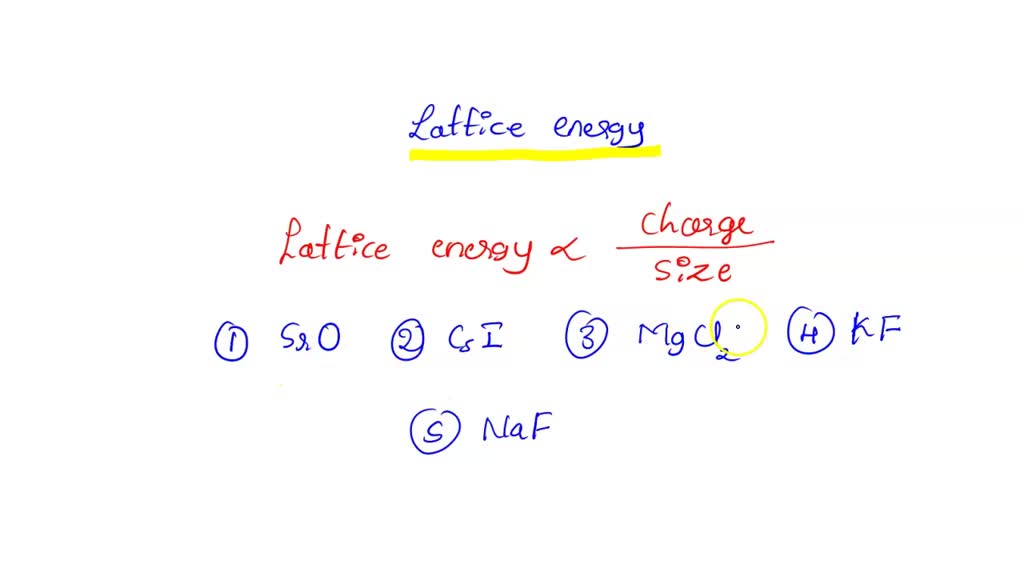
SOLVED: Based on ionic radii and charges, which of the following solids would have the greatest lattice energy? SrO CsI MgCl2 KF NaF